[row][column][textblock]
Volume 7 Issue 4
Lisa DeVetter (Assistant Professor, Dept. Horticulture at WSU), Beverly Gerdeman (Assistant Research Professor, Dept. Entomology at WSU), Matt Arrington (Former PhD Graduate Student, Dept. Horticulture at WSU), Hollis Spitler (Agricultural Technician, Dept. of Entomology at WSU), Bill Snyder (Professor, Dept. of Entomology at WSU), and Olivia Smith (PhD Student, Dept. of Biology at WSU)
[/textblock][/column][/row][row][column][textblock]Cool temperatures and precipitation during bloom limit honey bee foraging, which can reduce pollination and subsequent yields in blueberry. The shape of blueberry flowers and their low nutritional quality also contribute to making them difficult to pollinate and uninviting to honey bees. Scientists have been looking at alternative ways to improve pollination and increase blueberry yields. Michigan State University researchers Drs. Rufus Isaacs and Brett Blaauw have found that hedgerows and flower strips can promote pollination in blueberry by increasing pollinator diversity and abundance. In these trials, they found an increase in beneficial insects along with an increase in fruit set, berry weight, and seed number/berry in fields next to established wildflower plantings compared to those adjacent to mowed strips. These findings suggest similar benefits may be achieved through border vegetation management in Washington blueberry fields.
Despite these encouraging findings, there is concern regarding the potential negative aspects of border vegetation on blueberry production. Arborvitae (Thuja sp.) is a popular hedgerow species bordering blueberry fields in western Washington and functions to prevent pesticide drift onto adjacent properties. The primary insect pest targeted by spray programs is spotted wing drosophila (Drosophila suzukii; SWD), a direct pest whose larvae feed on developing fruit. Infestations of SWD lead to unsellable fruit or fruit that is downgraded based on poor quality. SWD overwinter in evergreen vegetation (Caprile et al., 2016) and have been recovered by Gerdeman in arborvitae, raising concern that border vegetation may serve as protective overwintering sites for SWD and encourage their establishment. Additionally, native flowering plants bordering blueberry fields may supplement SWD nutrition, enhancing establishment and increasing pest populations earlier in the season. Laboratory studies by Tochen et al. (2016) determined access to flowering cherries and blueberries boosted nutrient levels with the potential to increase SWD survivorship. Another concern is that border vegetation could provide habitat and increase populations of birds that feed on and damage blueberries, as well as transmit foodborne pathogens (e.g., human-pathogenic strains of E. coli and Salmonella) when they defecate in fields (Nielsen et al., 2004). Conversely, border vegetation could increase habitat for insectivorous birds that contribute to biological control of pest insects and of raptors (e.g., hawks, falcons) that kill pest birds (Boesing et al., 2017; Kross et al., 2012; Mäntylä et al., 2011; Symondson et al., 2002). The purpose of our project was to explore impacts of various adjacent border vegetation on several aspects of blueberry production in western Washington blueberry fields.
To achieve this, we measured pollination services, populations of beneficial and pest insects and bird species, and production attributes of blueberry (‘Duke’, ‘Draper’, and ‘Liberty’ ) grown next to 3 different types of existing border vegetation on 9 commercial farms in Whatcom and Skagit counties, Washington in 2017. Our border vegetation treatments include: 1) Control (primarily medium-height grasses); 2) Woody perennial vegetation [mixture of woody species including Cedar (Cedrus sp.) and Arborvitae (Thuja sp.)]; and; 3) Herbaceous vegetation [mixture of monocots (e.g., Poa sp. and quackgrass (Elymus sp) and broadleaves (e.g., Taraxacum officinale)] (Fig. 1).[/textblock][/column][/row][row][column][textblock]
Figure 1. Border vegetation treatments adjacent to highbush blueberry plantings include: 1) Control (left); 2) Herbaceous vegetation (center); and 3) Woody perennial vegetation (right).[/textblock][/column][/row][row][column][textblock]Our study did not find any significant differences in honey bee (Apis mellifera) foraging activity (all sites were stocked at the same honey bee hive density) or bee species (primarily Bombus sp.) between the treatments.
To measure beneficial and pest insect species, weekly samples were collected with an insect vacuum (D-vac, Model-122) and apple cider vinegar traps were used to assess SWD. Results indicated 75% of arthropods collected were from the herbaceous and control borders (40% and 35%, respectively) compared with only 25% from perennial sites (Fig. 2). Collections indicated parasitic wasps and spiders were the most numerous beneficial arthropods, while aphids and thrips were the pest species observed most frequently. No SWD were collected, which is reflective of the year and low SWD populations.
Total numbers of aphids and parasitoid wasps where closely aligned. Parasitic wasps increased with an increase in border plant diversity, perhaps because bio-diverse borders were more likely to have nectar and pollen sources which boost the wasps’ egg production. Spiders do not require nectar or pollen and were more common in the perennial borders which provided structure for their webs. Oribatida, a group of fungivorous mites which feed primarily on algae and lichen, were present in large numbers in the herbaceous (300), control (515), and perennial borders (825). Perennial sites, while not particularly diverse, provide a perfect substrate for growth of algae and lichen that oribatids could feed on.[/textblock][/column][/row][row][column][textblock]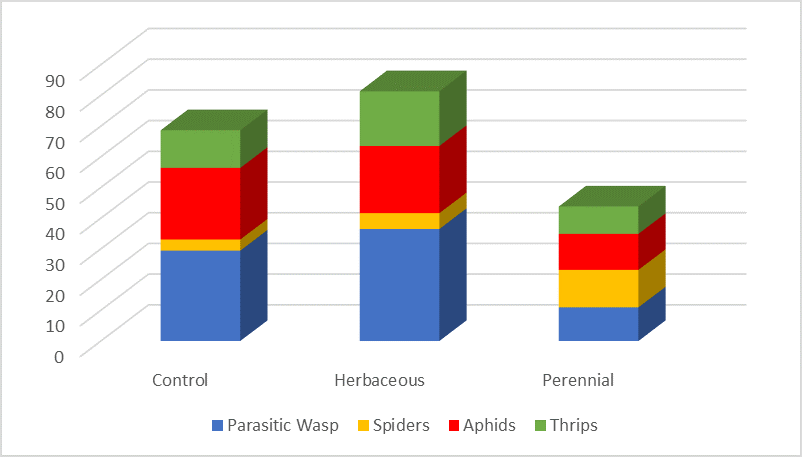
Fig. 2. Border site diversity and correlation with insect diversity. Y-axis represents total number of observed arthropods.[/textblock][/column][/row][row][column][textblock]The most significant finding was the absence of SWD at all locations. This highly unusual discovery follows overall trends observed in 2017 in much of western Washington. Total numbers of insects collected were low when sampling began in mid-May until mid-June, when insect populations showed a sharp increase for all three border types. No cultivar differences in pests and beneficials or timing of their appearance were detected between ‘Duke’ (early season), ‘Draper’ (mid-season), and ‘Liberty’ (mid-late season) despite variations in ripening of the berries. The increase in insect populations observed in mid-June across all three cultivars suggests very little benefit is derived from implementing one of the specific border vegetations under investigation. The weekly fluctuations observed in arthropod populations after mid-June most likely are responses to the weekly SWD insecticide applications, which begin as blueberries begin to ripen (data not presented). Despite lack of SWD in the blueberry fields or borders, growers are reluctant to reduce their weekly calendar SWD management programs due to the industry zero policy for SWD larvae in fruit.
Yellow sticky cards placed inside blueberry fields (in-plot) and on their perimeter (border vegetation) also detected greater populations of aphids and parasitic wasps in border vegetation but no significant differences between the treatments (Fig. 3). Populations increased between May and July as above.[/textblock][/column][/row][row][column][textblock]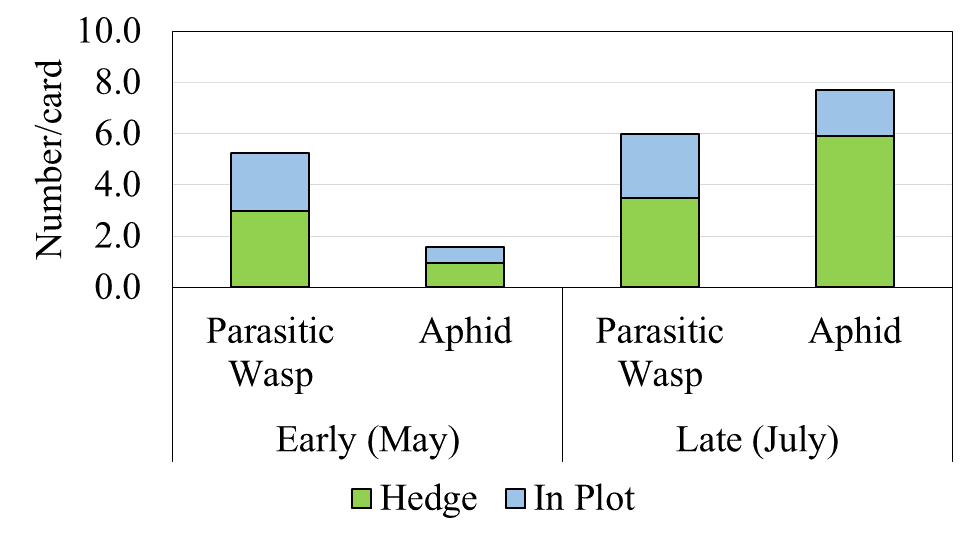
Figure3. Populations of parasitic wasps (beneficial) and aphids (pest) in vegetative borders (“hedge”) and within blueberry fields (“in-plot”) in May and July 2017.[/textblock][/column][/row][row][column][textblock]Our bird surveys (Figs. 4 and 5) indicated that the density of observed species was low, which is likely due to the use of bird deterrents observed across farms including cannons and “bird scarers”. House Finch (HOFI), which feed almost exclusively on plant material including seed buds and fruits, had the greatest average population density in our plots but did not appear to be influenced by border vegetation. American Robin (AMRO) and Cedar Waxwing (CEDW) were consistently observed within and beyond our survey plots but less often in the perennial habitat where insects were less abundant. American Crow (AMCR) and CEDW were observed more often outside plots rather than inside. White-crowned Sparrows (WCSP) were more commonly observed inside than outside plots, but few were observed inside our plots for the control treatment. Barn Swallow (BARS) had a greater average population density in the control habitat likely due to the presence of structures (barns, silos and wiring) providing perching and nesting sites. BARS also prefer open habitats relative to forested areas. Few raptors were observed, and most surprisingly no starlings were observed during the study.[/textblock][/column][/row][row][column][textblock]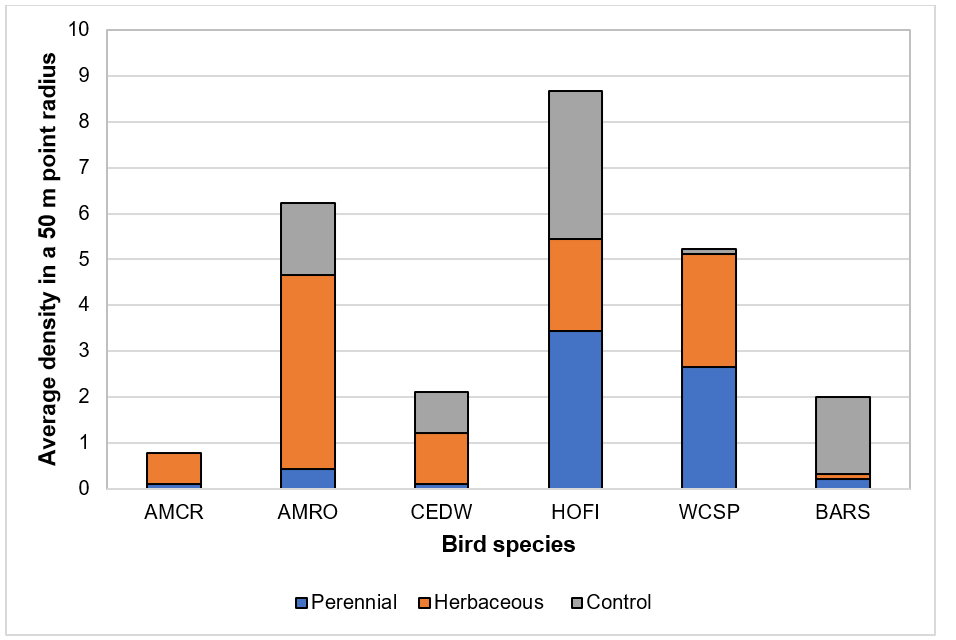
Figure 4. Average density by bird species and border vegetation treatment within a plot [plot = 50 m (164 ft) point radius], 2017. Abbreviations along x-axis are bird species, with AMCR = American Crow, AMRO = American Robin, CEDW = Cedar Waxwing, HOFI = House Finch, WCSP = White-crowned Sparrow, and BARS = Barn Swallow.
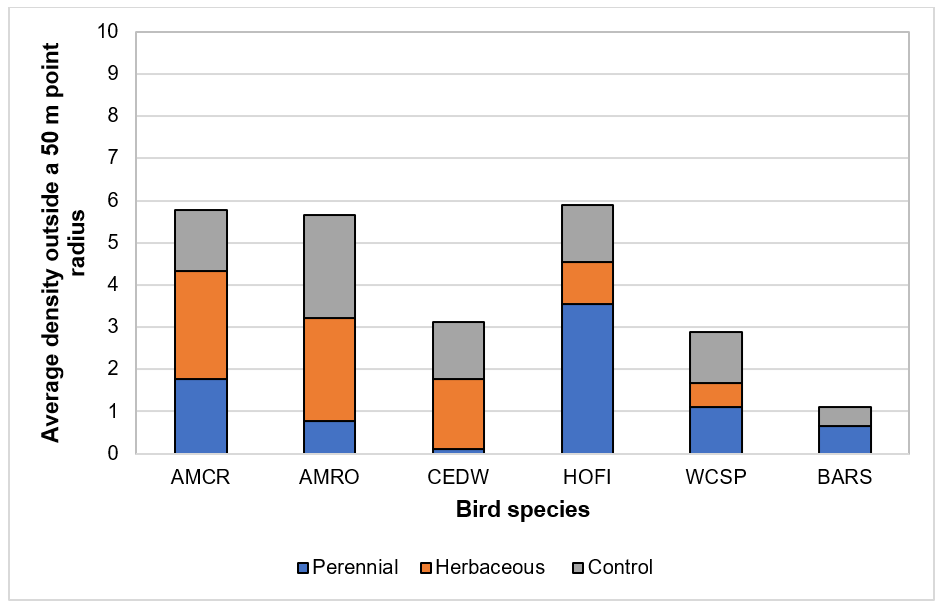
Figure 5. Average density by bird species and border vegetation treatment beyond a plot [plot = 50 m (164 ft) point radius] and as far a distance that could be observed (~150 m), 2017. Abbreviations along x-axis are bird species, with AMCR = American Crow, AMRO = American Robin, CEDW = Cedar Waxwing, HOFI = House Finch, WCSP = White-crowned Sparrow, and BARS = Barn Swallow.[/textblock][/column][/row][row][column][textblock]No differences in fruit set, berry weight, seed number, nor estimated yields were observed across treatments. Preliminary data from this project show that our border treatments have small to negligible impacts on our measured variables. Interestingly, we observed declines in arthropod populations that coincided with pesticide applications. Thus, in-field management practices, namely pesticide applications, appear to be driving arthropod diversity, not border vegetation. Despite these results, we did observe slightly greater arthropod diversity in our herbaceous border habitat treatment. This mirrors the work performed by Blaauw and Isaacs (2014 and 2015) in Michigan where diverse border habitats were associated with greater numbers of beneficial arthropods than pests. In their case, borders were intentionally seeded with wildflowers providing not only increased nectar and pollen, but the waist-high plants also provided protection. Intentionally planting flowering borders may increase arthropod diversity, but as long as SWD is managed with weekly sprays, any benefits will likely be negated. Currently growers will likely maintain their weekly SWD spray programs to ensure larvae-free berries until permanent changes in SWD populations are observed.
Acknowledgements
Funding for this project was provided by WSU’s BIOAg program. Thanks to Sean Watkinson and Clara TeVelde for assistance with data collection. Also, thank you to our grower collaborators.
References
Blaauw and Isaacs. 2014. Flower plantings increase wild bee abundance and the pollination services provided to a pollination‐dependent crop. J. Appl. Ecol. 51(4): 890-898.
Blaauw and Isaacs. 2015. Wildflower plantings enhance the abundance of natural enemies in adjacent blueberry fields. Biol. Control 91: 94-103.
Boesing, A.L., E. Nichols, and J.P. Metzger. 2017. Effects of landscape structure on avian-mediated insect pest control services: a review. Landscape ecology 32(5):931-944.
Caprile et al. 2016. Phenology of spotted wing drosophila in the San Joaquin Valley varies by season, crop and nearby vegetation. Calif Agr. 70(1):24-31.
Kross, S.M., J.M. Tylianakis, and X.J. Nelson. 2012. Effects of introducing threatened falcons into vineyards on abundance of passeriformes and bird damage to grapes. Conservation Biology 26(1):142-149.
Mäntylä, E., T. Klemola, and T. Laaksonen. 2011. Birds help plants: a meta-analysis of top-down trophic cascades caused by avian predators. Oecologia 165(1):143-151.
Nielsen et al. 2004. Verocytotoxin-producing Escherichia coli in wild birds and rodents in close proximity to farms. Appl. Environ. Microb. 70:6944–6947.
Symondson et al. 2002. Can generalist predators be effective biocontrol agents? Annu. Rev. Entomol. 47:561–594.
Tochen et al. 2016. Impact of floral feeding on adult Drosophila suzukii survival and nutrient status. J. Pest Sci. 89:3:793–802.
[/textblock][/column][/row]