Volume 11 Issue 10
Purnima Puri1, Lisa Wasko DeVetter1, Chakradhar Mattupalli1
1 WSU Mount Vernon Northwestern Washington Research and Extension Center, Mount Vernon, WA 98273
[Funding for this research was provided by the Washington Blueberry Commission. Blueberry plants were generously donated by Dr. Claire Luby, USDA-ARS (Corvallis, OR) and Fall Creek Farm and Nursery, Inc., (Lowell, OR)]
Problem:
- Mummy berry, a fungal disease caused by Monilinia vaccinii-corymbosiis one of the major threats to blueberry. The disease is especially challenging to manage organically and has the potential to cause production losses up to 70%.
- Primary symptoms include blighting of young vegetative and floral tissues, sometimes referred to as “strikes” or “Monilinia blight”. Secondary infection occurs when pollinators (primarily honeybees) transfer the inoculum to open flowers. As the disease progresses, developing fruits turns reddish pink rather than blue. The fruits eventually become shriveled and mummified, which is why it’s called mummy berry disease. Deformed fruits are non-acceptable in both fresh and processing markets.
- Mummified fruits that fall on the ground become the inoculum source for the next season.
- Mummy berry management is primarily driven by calendar-based application of fungicides but developing disease-resistant or low susceptibility cultivars would be an optimal long-term solution.
Objective:
- Develop and implement a mummy berry screening protocol that can be used to select blueberry genotypes with resistance or low susceptibility to the disease
Approach:
- Five container-grown plants of ten blueberry genotypes (‘Aurora’, ‘Bluejay’, ‘Calypso’, ‘Duke’, ‘Liberty’, ‘Northland’, ORUS264-1, ORUS292-2, ORUS587-1, and ORUS620-2) were maintained in a screenhouse at the WSU Mount Vernon Northwestern Washington Research and Extension Center (Figure 1).
- Inoculum was prepared by dislodging conidia from blighted leaf tissues collected from local infested fields. Inoculations were performed manually by applying a known concentration of the inoculum (5.75 x 105 conidia/mL) in suspension to the stigma of open flowers using a paintbrush. Following inoculation, freshly collected pollen was applied to the stigma of the flower using a paintbrush to ensure adequate pollination and fruit development.
- Inoculations and pollinations were carried out on different dates (4th, 9th, 10th, and 27th May 2022) coinciding with the development of new flowers. Following inoculation, unopened flowers in the flower cluster were removed and a ribbon was tied to the shoot to differentiate the inoculated flower cluster as well as ensuring the flowers were inoculated only once.
- Green fruits from inoculated plants were collected on 3rd and 5th of July 2022 and cut transversely to examine the presence of fungal growth (Figure 1).





Preliminary results:
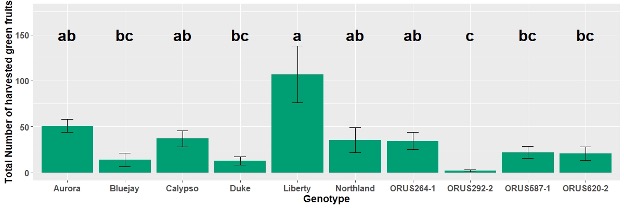
- As blueberry plants were obtained from various sources, plant age differed resulting in differences in the total number of green fruits harvested from each genotype (Figure 2). Data analysis was conducted after square root transformation.
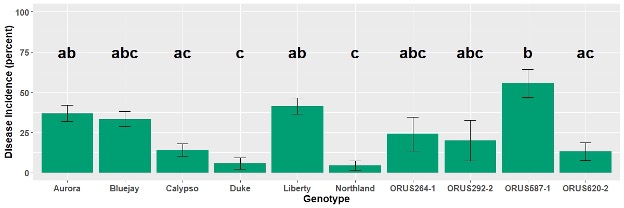
- Disease incidence was calculated based on the number of green fruits showing pathogen signs and the total number of harvested green fruits from each plant.
- A non-parametric test was conducted to analyze the data and differences were observed among genotypes for mummy berry fruit rot incidence (Figure 3).
- Mummy berry fruit rot incidence was lowest in ‘Northland’ and ‘Duke’, and significantly different from ORUS587-1.
Conclusions and future work:
- Differential responses among genotypes were observed for mummy berry fruit rot incidence indicating that the current experimental procedures are useful for continued genotypic screening for resistance or low susceptibility to mummy berry fruit rot.
- In addition to fruit rot evaluations, future experiments will include screening for mummy berry shoot blight incidence.